COVID-19 Antigen Self-Test von Siemens Healthineers erhält in Deutschland Sonderzulassung für Eigenanwendung durch Laien – GHA – German Health Alliance

Siemens Healthineers Announces CE-Mark for CLINITEST Rapid COVID-19 Antigen Self-Test for Unsupervised Self Testing

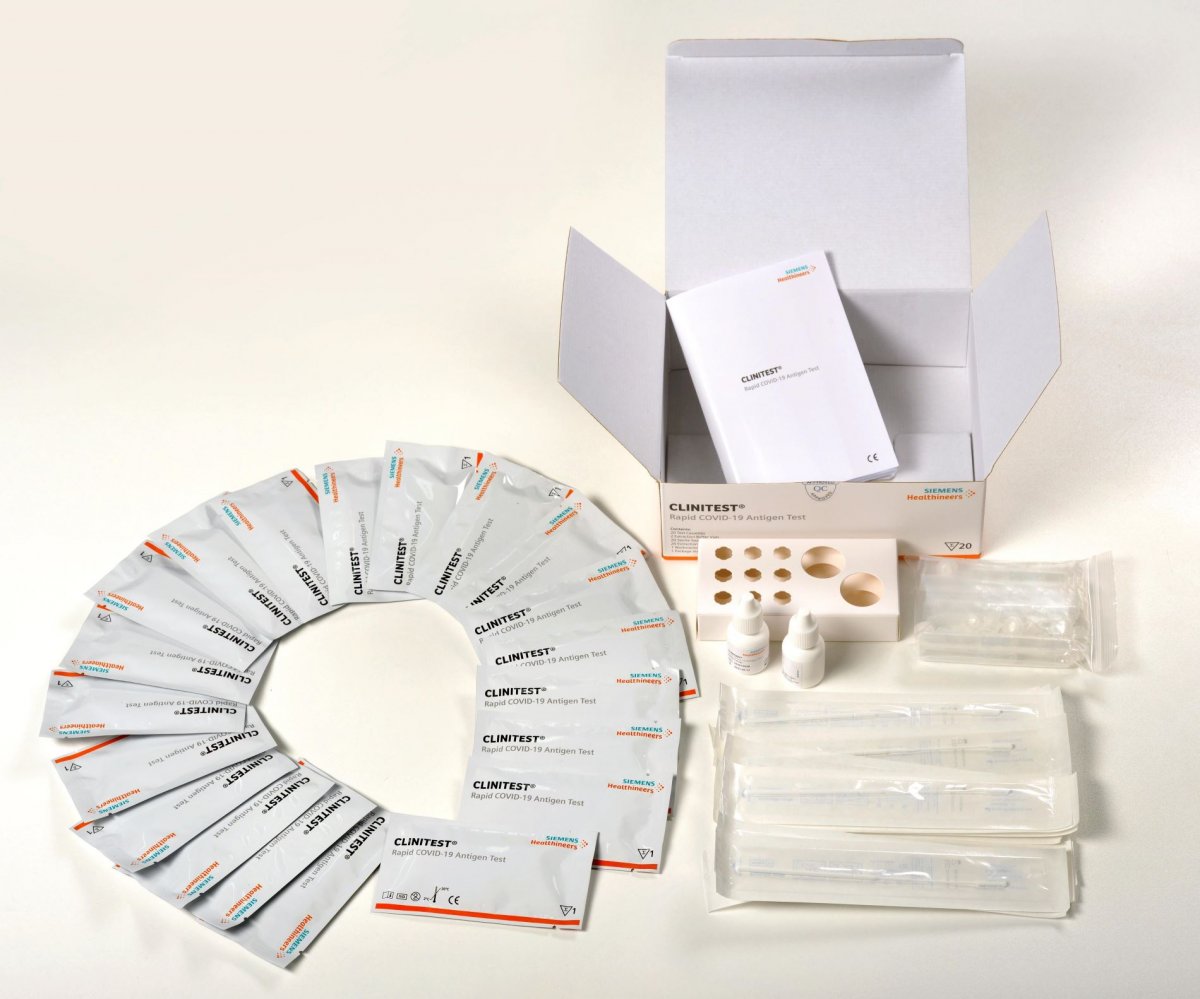
Amazon.com: CLINITEST Rapid COVID-19 Antigen Self-Test, 1 Pack, 5 Tests Total, Test results in 15 minutes, FDA EUA Authorized OTC at-Home Self Test Kit : Industrial & Scientific
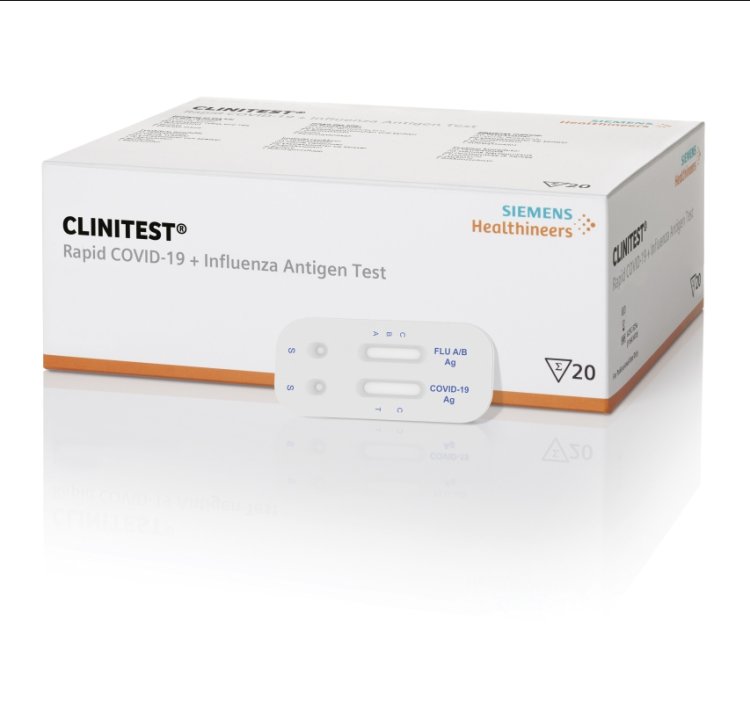
Siemens Launches New Combination Tests for SARS-CoV-2 and Seasonal Respiratory Pathogens - COVID-19 - Labmedica.com

Amazon.com: CLINITEST Rapid COVID-19 Antigen Self-Test, 1 Pack, 5 Tests Total, Test results in 15 minutes, FDA EUA Authorized OTC at-Home Self Test Kit : Industrial & Scientific

CLINITEST Rapid COVID-19 Antigen Self-Test | United States of America | CLINITEST Rapid COVID-19 Antigen Self-Test has received FDA Emergency Use Authorization in the United States. https://bit.ly/3zdnMyX | By Siemens Healthineers

Siemens Healthineers CLINITEST Rapid COVID-19 Antigen Self-Test Nasal Swab:Diagnostic | Fisher Scientific

Diagnostic accuracy of covid-19 rapid antigen tests with unsupervised self-sampling in people with symptoms in the omicron period: cross sectional study | The BMJ